Biopharmaceutical News week 6 2015
Acquisitions /mergers/joint-ventures
February 2, 2015
Icon buys MediMedia Pharma Solutions for $120 million
Icon (Ireland) is to acquire MediMedia Pharma Solutions (Yardley, USA) for $120 million, creating “the industry’s leading integrated scientific communications and market access solution". Icon chief executive Ciaran Murray said “the need to demonstrate and communicate the value of new medicines is an important global healthcare trend” and the acquisition of MediMedia Pharma Solutions “deepens the expertise” of the company’s existing operations “and brings us outstanding scientific and medical communications capabilities”.
February 5, 2015
Pfizer acquires Hospira for $17 Billion
Hospira (Lake Forest, USA) will be acquired by Pfizer for about $17 billion in cash. Hospira is known for its business in sterile and generic injectables, devices and biosimilars. Hospira teamed up with Celtrion (South Korea) to be early in the game of the biosimilar wave. It already markets biosimilars of two Amgen drugs Retacrit and Nivestim, outside the U.S., and recently won European approval for its J&J’s Remicade biosimilar.
Business
January 30, 2015
Ortho-Clinical Diagnostics signs transfusion diagnostics deal with Quotient
Ortho-Clinical Diagnostics signs an exclusive agreement with Quotient and will pay $69 million for the rights to market MosaiQ, its transfusion diagnostics platform.
February 1, 2015
Novartis faces suspension in Japan
According to the Japan Times, the Ministry of Health, Labour and Welfare's plans to suspend Novartis ‘s pharma business order for about 15 days, for repeated violations for failing to report adverse events promptly. The company was already facing charges of exaggerated advertising while promoting its antihypertensive drug Diovan. In addition a scandal erupted in Spring of last year, when Novartis failed to report side effects of Gleevec and was accused to break protocols in a clinical trial of the leukemia drug Tasigna.
Approval of drugs, vaccines, diagnostics and devices
January 29, 2015
US Senators seek to modernize NIH and FDA to expedite approval of new treatments
US Senators Lamar Alexander (Tennessee), and Richard Burr (North Carolina) presented their plans to modernize the FDA and the NIH as part of efforts to expedite the development and approval of new treatments.
February 3, 2015
US FDA releases draft guidance on biosimilar applications
The FDA has released draft guidance in biosimilar marketing applications submitted through the 351(k) pathway. According to the draft, applications should include data showing that a biosimilar is interchangeable and can generate the same clinical result as its reference biologic in all patients. The application, or a supplement to the application, should include the data demonstrating biosimilarity. Drugmakers can comment on the draft through April 6.
February 2, 2015
US FDA approves diabetes combination drug from Lilly and Boehringer
Partners Eli Lilly and Boehringer Ingelheim gain FDA approval for Glyxambi, a diabetes treatment combining two existing drugs into a single tablet. Glyxambi, is a fixed dose combination of empagliflozin, a sodium glucose co-transporter-2 inhibitor, and linagliptin, a dipeptidyl peptidase-4 inhibitor, in a once-daily tablet. It is the first such combination approved by the FDA.
February 2, 2015
Sanofi launches Affreza the only inhaled insulin available in U.S.

Afrezza-inhaled-insulin-drug-device- MannKind
After the failure of Exubera (Pfizer) several years ago, Sanofi and MannKind, which developped the drug, are launching Affreza, the first inhaled insulin in the US. Afrezza is priced at $7.54 for a daily dose of 12 units compared to $3.14 for Sanofi's injectable Apidra. MannKind executive Alfred Mann said he's "proud" that the long, hard R&D journey is at an end. And Sanofi's Pierre Chancel, SVP of its diabetes division, called Afrezza "an important addition to Sanofi's growing diabetes portfolio."
February 3, 2015
Merck launched new first-in-class sleep drug in the US
Almost twenty five years after the approval of Stilnox/Ambien (zolpidem) by Searle and Sanofi, Merck launches a novel treatment for sleep disorders with Belsomra (suvorexant).The drug is the first orexin receptor antagonist approved for the treatment of insomnia. This new class of medicines works by blocking the action of orexin, one of the neurotransmitters involved in wakefulness . The drug is classed a Schedule IV controlled substance because it can be abused or lead to dependence.
February 4, 2015
TissuGlu from Cohera gets first FDA approval of a tissue adhesive for internal use

TissuGlu-technology-Cohera-Medical
US FDA allows Cohera Medical Inc. (Pittsburgh, USA) for use of the urethane-based adhesive TissuGlu the first tissue adhesive approved for internal use during abdominoplasty surgery. TissuGlu allows for the elimination of external drains after surgery. TissuGlu received a CE mark in 2011. More than 1,500 surgical procedures have used the product. TissuGlu is an adhesive consisting of a polyurethane pre-polymer which polymerizes at the surgical site upon contact with moisture and is delivered via a device that contains 5 milliliters of the adhesive.
US FDA approves Medtronic’s Pipeline Flex device for brain aneurysm
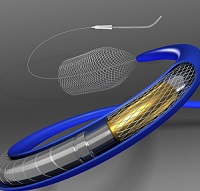
Pipeline-Flex-High-Medtronics
Medtronic received its first new FDA approval resulting from the acquisition of Covidien. The approval is for Pipeline Flex, a minimally invasive flow diversion device for unruptured aneurysm. It received a CE mark and was launched in Europe earlier last year. The Pipeline Flex has a braided cylindrical mesh tube that is implanted across the base or neck of an aneurysm. It cuts off blood flow to the aneurysm, thereby reconstructing the diseased section of the blood vessel. It is specifically intended for the endovascular treatment of complex intracranial aneurysms.
February 4, 2015
US FDA approves Pfizer's novel breast cancer drug Ibrance
US FDA regulators have granted accelerated approval to Pfizer’s first-in-class cyclin-dependent kinase 4/6 inhibitor Ibrance (palbociclib) for the treatment of postmenopausal women with certain forms of advanced breast cancer. Backed by impressive progression-free survival data, the drug has been approved for use alongside letrozole to treat postmenopausal women with oestrogen receptor-positive, human epidermal growth factor receptor 2-negative (ER+/HER2-) advanced breast cancer.
Drugs at clinical stage
3 FDA Breakthrough Therapy Designations: Intercept Pharmaceuticals, Genentech and Bluebird Bio
Three FDA Breakthrough Therapy Designations (BTDs) are announced in the past week by sponsor companies:
- Intercept Pharmaceuticals, on January 29th, for Obeticholic acid, a first-in-class farnesoid X receptor agonist, as a treatment for nonalcoholic steatohepatitis (NASH) with liver fibrosis.
- Bluebird, on February 2, a gene therapy for transfusion-dependent patients with beta-thalassemia.
- Roche and Genentech, on February 2, for experimental immunotherapy MPDL3280A, for use in patients with PD-L1 (programmed death-ligand 1) positive non-small cell lung cancer.
Technology Guardant Health expands Guardant360, its liquid biopsy cancer test
Guardant360 is a cancer blood test with a 68-gene panel available now in the U.S. It offers a comprehensive cancer view of tumor genomic alterations in real time. The test is intended as a complement to traditional biopsy. Patients who would benefit most from using it are those for whom a tumor biopsy is not an option, those who failed treatment, and/or patients with metastatic cancer.
Mobile devices may radically change healthcare
Today's medical practice will undergo a radical transformation as mobile devices become more Health-friendly, according to Eric Topol, director of the Scripps Translational Science Institute in San Diego. “It will take a new plasticity of the medical community in facing its greatest and singular challenge since the profession's origin–its transformation by pervasive embracement of digital technology," Topol and Scripps colleagues write. Their expectations are supported by analyst Mike Feibus, who said that “the transition shift of mHealth technology from the fitness realm to the medical and home patient monitoring environments will take root this year”.
Miscellaneous
Obama proposes $215M for personalized medicine initiative
US President Barack Obama will ask Congress for $215 million to fund his precision medicine initiative. Under the proposal, $130 million would go to the National Institutes of Health (NIH) to support development of a national cohort for research and $70 million would go to National Cancer Institute (NCI) research. The FDA would get $10 million to support database development, and the Office of the National Coordinator for Health IT would get $5 million to advance interoperability and secure data exchange. An earlier White House Statement said "most medical treatments have been designed for “the average patient”….”as a result treatments can be very successful for some patients, but not for others."
February 3, 2015
UK: The place to be for life sciences investors?
At the inaugural Future of Healthcare Investor, hosted by the London Stock Exchange and MedCity, Cathrin Petty, co-head of healthcare investment banking EMEA at JP Morgan, said the UK “has all the right ingredients to create a successful and sustainable life science market: the science, the clinical development infrastructure and management. We now need the long term capital to fund it”. Last year saw £1.25 billion raised in life science IPOs and follow-on offerings on the London Stock Exchange, the most in this sector since 2007.
US reports a sharp drop in breast cancer deaths over the past 20 years
According to a USA Today report, the past 20 years have seen a sharp drop in the number of deaths from breast cancer in the U.S. The number of deaths fell from 33 to 22 per 100,000 women between 1990 and 2011. This decline represents more than 200,000 lives saved, the American Cancer Society said. Experts estimate that the trend has continued in the past four years as well, and can be attributed to better treatment, rising awareness and more frequent checkups.
February 6, 2015
Margaret Hamburg resigns as head of FDA
After several weeks of rumors Dr. Margaret Hamburg has confirmed that she is stepping down from her position of commissioner of the US Food and Drug Administration.
Author : Jean-Claude MULLER, Special Advisor, Innovation & International Relationship (I&IR)
Discover our services in Marketing & Business Development:
See All News
See other Biopharmaceutical News
see other Pharma & Biotech events in 2014
1
Last News
- US FDA Novel Drug Approval for 2025
- Update on Alzheimer’s disease treatments
- Le Populisme en France : Une note d’éclairage
Events
News archives
- January 2026
- December 2025
- November 2025
- September 2025
- July 2025
- June 2025
- May 2025
- April 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- March 2024
- February 2024
- January 2024
- November 2023
- September 2023
- July 2023
- April 2023
- March 2023
- January 2023
- December 2022
- November 2022
- October 2022
- August 2022
- June 2022
- May 2022
- April 2022
- March 2022
- January 2022
- December 2021
- November 2021
- October 2021
- September 2021
- August 2021
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- October 2018
- June 2018
- May 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- September 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- August 2015
- July 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- June 2014
- May 2014
- April 2014
- March 2014
- January 2014
- November 2013
- September 2013
- July 2013
- May 2013
- April 2013
- March 2013
- January 2013
- December 2012
- November 2012
- October 2012
- March 2012





