Biopharmaceutical News Week 5- 2016

We wish all our Chinese friends a happy, healthy and prosperous New Year of the Monkey!
“Kung Hei Fat Choy!”
Zika virus
The World Health Organization (WHO) declared a global emergency over the explosive spread of the Zika virus (ZIKV). The last time WHO declared a public health emergency was for the Ebola outbreak in West Africa, which killed more than 11,000 people. "After a review of the evidence, the committee advised that the clusters of microcephaly and other neurological complications constitute an extraordinary event and public health threat to other parts of the world," WHO Director-General Dr. Margaret Chan said. WHO estimates there could be up to 4 million cases of Zika in the Americas in the next year, but no recommendations were made to restrict travel or trade. Nevertheless the Centers for Disease Control and Prevention (CDC) have issued travel recommendations for pregnant women to post-pone travel to countries in Latin America and the Caribbean where ZIKV transmission is ongoing.
Sanofi Pasteur (Lyon and Paris, France) has announced that it has launched a vaccine research and development project targeting the prevention of the Zika virus infection. This makes Sanofi the first major pharmaceutical company initiate such a project. Sanofi Pasteur's expertise with established R&D and industrial infrastructure for the newly licensed vaccine for dengue, Dengvaxia®, can be rapidly leveraged to help understand the spread of ZIKV and potentially speed identification of a vaccine candidate for further clinical development. NewLink Genetics (Ames, Iowa, USA) as well as the UK Medical Research have also announced the start of their own program.
The Centers for Disease Control and Prevention (Atlanta, USA) has reported the first known case of Zika virus transmission in the US (Dallas, Texas) and confirmed that it was contracted through sex and not a mosquito bite. Up to now it was thought that Zika was spread only by mosquitoes of the Aedus genus; if sexual contact is confirmed as a mode of transmission the spread of the disease will become far more alarming.
In addition to the US, Ireland has reported its first two cases, following one case in Denmark, six in the UK, two in Australia and one pregnant woman in Spain.
Acquisitions /mergers/joint-ventures
ChemChina (Beijing, China) is offering $43 billion to acquire Syngenta (Basel, Switzerland) in a deal that would be the largest ever takeover by a Chinese company. If the transaction is complete ChemChina will become the world largest supplier of pesticides and agrochemicals, ahead of Monsanto, Dow and Bayer Crop Science.
According to well-informed sources close to the business, Sanofi is considering stepping out of the 21-year partnership it has established with Merck in their Sanofi MSD joint venture based in Lyon (France). The two companies own equal stake in Sanofi Pasteur MSD, with reported sales of around $330 million in 2015, and which markets in Europe the second and third-best selling vaccines worldwide: Gardasil and Fluzone/Vaxigrip.
Abbott acquires Alere (Waltham, MA, USA) for $5.8 billion and becomes the world’s premier point-of-care testing business, including products for infectious disease, molecular, cardiometabolic and toxicology testing and strengthens Abbott’s diagnostics presence with benchtop tools and rapid strip tests.
Stryker (Kalamazoo, MI, USA) acquires Sage Products (Cary, IL, USA), a disposable device maker, for $2.775 billion and expects to significantly reduce hospital acquired infections. The move demonstrates that reducing hospital-acquired infections has become a top priority for med tech players mainly due to US federal government incentives. For instance, a section of the Affordable Care Act (ObamaCare) mandates a 1% Medicare payment reduction for hospitals that fall in the top 25% in certain hospital-acquired infections.
Business
Portola Pharmaceuticals (South San Francisco) licenses Japan rights of andexanet alfa, an antidote to anti-coagulants apixiban and other Factor Xa inhibitors to Bristol-Myers Squibb and Pfizer. Under the terms of deal with BMS and Pfizer, Portola will receive an upfront payment of $15M, milestones up to $90M and double-digit royalties
…and announces a clinical collaboration for Japan with Bayer Healthcare to include its own Factor Xa inhibitor rivaroxaban. Under the terms of the deal, Bayer will pay an upfront payment of $5M and an undisclosed milestone based on regulatory approval. Three Factor Xa inhibitors are currently available in Japan, BMS and Pfizer’s apixaban, Bayer's rivaroxaban and Daiichi Sankyo's endoxaban but no antidote is approved yet.
GlaxoSmithKline is expanding its immunotherapy alliance contracted last June with Adaptimmune Therapeutics (Philadelphia, PA, USA and Oxford, UK) to accelerate GSK337779, an enhanced T-cell drug that targets NY-ESO-1, one of the best-characterized and most immunogenic cancer antigens, into pivotal clinical trials for synovial sarcoma and explore the treatment of liposarcoma as well as eight combinations with other therapies, including checkpoint inhibitors. Under the modified deal, GSK will pay up to $500 million in milestone payments if the company successfully develops the treatment for at least two disease indications and at least two human leukocyte antigen types.
Teva enters into an antibody deal with AbCellera (Vancouver, Canada) to discover rare monoclonal antibodies using AbCellera’s high throughput single cell antibody platform. Under the terms of the deal Teva will pay an undisclosed upfront payment, research payments, as well as contingent development and regulatory milestones. Last week AbCellera announced that it will be collaborating with Merck & Co. to generate antibodies against an undisclosed disease target.
Takeda Pharmaceuticals extends again its partnership with Mersana Therapeutics (Cambridge, MA, USA) and gets rights, out of the US and Canada, to XMT-1522, a Fleximer-based antibody-drug conjugate that targets HER2+ expressing tumors in breast, gastric and non-small cell lung cancers and to use the Fleximer technology to research novel compounds. The agreement broadens the ongoing collaboration started in 2014. Under the terms of this deal, Takeda will pay a $40 million upfront payment and about $750 million in milestone fees plus sales royalties, and an additional payment of $20 million contingent on the approval of the drug's IND.
Approval of drugs, vaccines, diagnostics and devices
US FDA approves:
Avanir Pharmaceutical’s (Aliseo Viejo, CA, USA) Onzetra, an intranasal candidate for the treatment of migraines. The drug had been rejected by the FDA in 2014. The Xsail Breath Powdered Delivery Device will administer 22 milligrams of a dry powder formulation of sumatriptan, the most commonly prescribed migraine drug.
… and a single-dose version of Merck’s Emed (fosaprepitant dimeglumine) in combination with other anti-emetics, for the prevention of delayed nausea and vomiting in adults taking chemotherapy. The approval makes Emend the first intravenous single-dose NK1 receptor antagonist approved in the US for both highly and moderate emetogenic chemotherapy.
European Commission approves AstraZeneca’s Tagrisso (AZD9291 or osimertinib, a third generation EGRF tyrosine kinase inhibitor) for the treatment of adult patients with locally advanced or metastatic epidermal growth factor (EGRF) T790M mutation positive non-small cell lung cancer. Tagrisso is the new first medicine to win clearance under the European Commission’s expedited process.
Drugs at clinical stage
US FDA grants:
breakthrough status to Allergan’s (Dublin, Ireland) raspastinel as an adjunctive treatment for major depressive disorders (MDD). Rapastinel (formerly known as GLYX-13) is a novel amidated tetrapeptide acting as a partial agonist of the glycine site of the NMDA receptor with a rapid onset of activity. The investigational intravenous formulation of raspatinel showed clear signs of efficacy one day after a single dose in a Phase II clinical trial of patients with MDD who had an inadequate response other antidepressants.
fast-track status:
- to Eagle Pharmaceuticals’ (Woodcliff Lake, NJ, USA) injectable suspension Ryanodex (dantrolene sodium) for the treatment of exertional heat stroke (EHS) a potential fatal condition. EHS is a rare, sudden and unpredictable disorder that can result in severe multi-organ dysfunction and death, characterized by core body temperature of 104° F (40° C) or greater and significant neurological dysfunction. The condition is more commonly seen in young people undergoing exertional physical activity in a hot weather environment, and is a leading cause of death in young athletes. Ryanodex would be the first medication for the potentially fatal condition.
- to Nimbus Therapeutics’ (Cambridge, MA, USA) NDI-010976 for the treatment of nonalcoholic steatohepatitis (NASH). NDI-010976 is an allosteric acetyl-coenzyme A carboxylase (ACAC) inhibitor that plays a key role in the metabolism of fatty acids.
- to TapImmunes’ (Jacksonville, FL, USA) TPIV, a multi-epitope folate receptor alpha peptide vaccine as a maintenance treatment for platinum-sensitive advanced ovarian cancer patients who achieved stable disease or partial response.
orphan status to Asterias Biotherapeutics’ (Freemont, CA, USA) AST-OPC1, an oligodendrocyte progenitor population derived from human embryonic stem cells, for use in patients with acute spinal cord injury. The activities of AST-OPC1 include production of neurotrophic factors, stimulation of vascularisation, and induction of remyelination of denuded axons, all of which are critical for survival, regrowth and conduction of nerve impulses through axons at the injury site
Science and Technology
The UK Human Fertilization and Embryo Authority (HFEA) decided to approve the first study in the world to use the new gene editing technique CRISPR to permanently change DNA in a human embryo. The study led by Kathy Niakan, a biologist at Francis Crick Institute, will attempt to edit out bits of DNA that prevent an embryo from developing properly—which may answer important questions about infertility. The embryo would not be allowed to survive beyond 14 days, meaning they wouldn’t be implanted into a woman’s womb and grown into live babies. We have described the technology and the ethical issues related to the technique in Biopharmaceutical NewsWeek 52-2015
There are many different ways to make nanomaterials but weaving has not been one of them until recently. Researchers led by Professor Omar Yhaghi at the U.S. Department of Energy Lawrence Berkeley National Laboratory and the University of California, Berkeley, have found a way to weave long-chain organic molecules into an interlacing pattern without irreversibly bonding them together. The resulting molecular “fabric”, which has unusual properties, could potentially lead to a variety of other materials with applications from porous crystals, polymer chemistry and drug delivery to information storage. Professor Jean-Pierre Sauvage from the University of Strasbourg (France), whose group developed the idea of templating around a metal ion, back in 1984, describes the work as “a great paper”. The article untitled “Weaving of organic threads into a crystalline covalent organic framework” was published in Science 22 JANUARY 2016, VOL 351, ISSUE 6271 page 365.
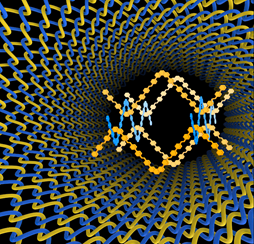
COF-505 is the first 3D covalent organic framework to be made by weaving together helical organic threads.
Medical Devices and Diagnosis News
Qiagen (Hilden, Germany) is teaming up with big data firm CosmosID (Aarhus, Denmark) to perform metagenomics microbial analysis. "Researchers running metagenomic, microbial and other applications often need to rely on complicated data analysis tools from different sources," SVP of Bioinformatics Laura Furmanski said in a statement.
Illumina (San Diego, CA, USA) extends its next-generation sequencing (NGS) technology to four separate institutions namely Vanderbilt University at Atlanta, University of Colorado at Denver, Partners HealthCare and Montreal Heart Institute, to facilitate genetic research and "accelerate discoveries that will improve human health," Illumina said in a statement. As a first step, scientists will use the company's sequencing systems to analyze 200,000 samples and probe genetics behind complex diseases such as Alzheimer's, bipolar disorder and Crohn's disease. Eventually, large sample collections at each research institution will be genotyped using Illumina's technology.
Cost, Pricing and Market Access
Roche does not expect its anti-cancer drugs to be caught in a short term US pricing pressure debate because drugs such as Rituxan, Avastin and Herceptin are treating patients with few other options according to Daniel O’Day Roche’s head of pharmaceuticals. "There will be a bifurcation of the industry. There will be true innovators that are providing transformational medicines. And then there will be the generic medicines," O'Day said, as quoted by Reuters.
At a highly-publicized hearing of the House Committee on Oversight and Government Reform on Capitol Hill in Washington DC, where recent drug-price hikes were on trial, Valeant interim CEO Howard Schiller and former Turing Pharmaceuticals CEO Martin Shkreli were asked questions on their drug pricing policy in the US. Martin Shkreli, who is out on a $5 million bail after being arrested in New York City in December on securities-fraud charges unrelated to the price increase, infuriated members of US Congress, when he refused to testify and pled the Fifth Amendment.Representative Elijah Cummings, the top Democrat on the House Committee, chastised Valeant and Turing for buying drugs and then raising prices for their financial gain. "They bought them, jacked up the prices, took as much money as they could out of the pockets of patients, hospitals and others, and then put those funds into their own coffers," Cummings said, as quoted by Bloomberg. Before the Congress hearing was over Shkreli tweeted "Hard to accept that these imbeciles represent the people in our government."
Miscellaneous
According to the French Institut de Veille Sanitaire (InVS), the life expectancy of French adults affected by prostate, breast and colon cancers has substantially increased over the past years. ”These tendencies are very encouraging” according to the authors of the report. Five years survival for prostate cancer patients has increased from 72% to 94% over the last twenty years. Five years survival for colon cancer patients has increased from 54% to 63% and breast cancer patient survival has increased from 80 to 87% over the same period. Unfortunately this is not true for lung cancer which remains the first cause of cancer mortality in men and the second in women.
Sanofi announced important job cuts with almost 600 in France. According to information released by union leader, Thierry Bodin, who met with Sanofi officials, 280 to 296 open R&D positions will not be filled and another 255 jobs mainly commercial operations (155) and corporate offices (100) will be removed. Sanofi officials assured the unions that production sites would not be closed and most of the affected employees will be offered "fully-funded" retirement packages.
US President Barack Obama is requesting an additional $755 million in funding for the 2017 FDA and the NIH budget proposal to support the Cancer MoonShot 2020 initiative he had announced during last month’s State of the Union address.
Bioevents
- BIO CEO Investor Conference on February 8-9 in New York City (USA)
- BIOCOM Global Life Science Partnering Conference on February 24-25 in Torrey Pines (USA)
- BIO Asia 2016 on March 15-16 in Tokyo (Japan)
- 9th European Life Science CEO Forum and Exhibition on March 15-16 in Zürich (Switzerland)
- Global Healthcare Forum on March 16-17 in Singapore (Singapore)
- BioPharma Asia Convention on March 22-24 in Singapore (Singapore)
- Annual China Healthcare Investment Conference on March 29-31 in Shanghai (China)
- BIO Europe Spring on April 4-6 in Stockholm (Sweden)
- BIO-IT World Conference & Expo’16 on April 5-7 in Boston (USA)
- BIO Vision on April 13-14 in Lyon (France)
- BIO Trinity on April 25-27 in London (UK)
- Bio€quity Europe 2016 on May 10-11 in Copenhagen (Denmark)
- Strategic Alliance Management Congress on May 16-18 in Philadelphia (USA)
- China BIO Partnering Forum on May 18-19 in Suzhou (China)
- BIO International Convention on June 6-9 in San Francisco (USA)
- BIO Pharm America on September 13-15 in Boston (USA)
- 7th Innovation Days on October 3-4 in Paris (France)
- BIO Europe on November 7-9 in Cologne (Germany)
- BIO Latam on November 29-30 in San Juan (Puerto Rico)
- Biofit 2016 on November 30-December 1 in Lille (France)
IMPORTANT NOTICE: This document has been prepared by btobioinnovation and is provided to you for information purposes only. The information contained in this document has been obtained from sources that btobioinnovation believes are reliable but btobioinnovation does not warrant that it is accurate or complete. The views presented in this document are those of btobioinnovation’s editor at the time of writing and are subject to change. btobioinnovation has no obligation to update its opinions or the information in this document.
Author: Jean-Claude Muller, Special Advisor at I&IR, jcm@btobioinnovation.com
Give us your feed-back on the biopharmaceutical News !
Discover our services in Marketing & Business Development:
See All News
See other Biopharmaceutical News
see other Pharma & Biotech events in 2016
2
Last News
- US FDA Novel Drug Approval for 2025
- Update on Alzheimer’s disease treatments
- Le Populisme en France : Une note d’éclairage
Events
News archives
- January 2026
- December 2025
- November 2025
- September 2025
- July 2025
- June 2025
- May 2025
- April 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- March 2024
- February 2024
- January 2024
- November 2023
- September 2023
- July 2023
- April 2023
- March 2023
- January 2023
- December 2022
- November 2022
- October 2022
- August 2022
- June 2022
- May 2022
- April 2022
- March 2022
- January 2022
- December 2021
- November 2021
- October 2021
- September 2021
- August 2021
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- October 2018
- June 2018
- May 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- September 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- August 2015
- July 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- June 2014
- May 2014
- April 2014
- March 2014
- January 2014
- November 2013
- September 2013
- July 2013
- May 2013
- April 2013
- March 2013
- January 2013
- December 2012
- November 2012
- October 2012
- March 2012





