Biopharmaceutical News Week 8

February, 19, 2015
Sanofi Appoints Olivier Brandicourt as Chief Executive Officer

Olivier Brandicourt joins Sanofi from Bayer HealthCare where he was Chairman of the Board of Management and a member of the Executive Council. Previously, Brandicourt held numerous positions of increasing responsibility within Parke-Davis/Warner-Lambert and Pfizer. A physician by training, Olivier Brandicourt's career includes several senior positions in Europe, Canada and the United States. Brandicourt will take up his new duties on April 2, 2015.
Acquisitions /mergers/joint-ventures
February 20, 2015
Panasonic Healthcare to buy Bayer diabetes device business for as much as $2.3B
According to a report from Bloomberg, Bayer is considering selling its diabetes devices business to Panasonic Healthcare, The price for the Bayer diabetes device unit is expected to be between €1 billion ($1.1 billion) and €2 billion ($2.3 billion).
Business
February 18, 2015
Actavis changes name to Allergan
Actavis, the Dublin based company, announced that it intends to use the Allergan name as its corporate name and for its global branded pharmaceutical portfolio, and will retain the Actavis name for select geographic regions and product portfolios.
February 18, 2015
Sanofi signs agreement with Dutch Lead Pharma to develop autoimmune diseases drugs
Sanofi announced that it has entered into a research collaboration and license agreement with Dutch privately owned drug biotech discovery company Lead Pharma (located in Nijmegen and in Oss) to discover, develop and commercialize small-molecule therapies directed against the nuclear hormone receptors called ROR gamma t. This nuclear retinoic acid receptor-related orphan receptor gamma is a key regulator of the cytokine immune pathway, interleukin-17 (IL-17) and plays a critical role in a broad range of autoimmune disorders, including rheumatoid arthritis, psoriasis and inflammatory bowel disease.
Boston Scientific settles breach-of-contract Guidant suit with Johnson & Johnson
Boston Scientific settles its ongoing breach-of-contract lawsuit with Johnson & Johnson over a failed bidding for Guidant, for $600 million in 2006. The Massachusetts based company will pay far less than the $7.2 billion originally requested by J&J, and will not have to acknowledge any liability by Guidant. J&J sued Guidant, Abbott and Boston Scientific for breach of contract, claiming that Guidant told Abbott about the asset deal.
February 18, 2015
William Demant, hearing aid maker, attempting to buy French Audika for $191M
Denmark's William Demant, the world's second largest hearing aid maker, announced it is in talks to acquire French hearing correction provider Audika for €168 million ($191 million). The French company has 460 outlets in France and reported revenues of €98.7 million last year. At a price of €17.78 per share, the acquisition values Audika at a premium of 32%.
Rainbow Medical raises $25M from Chinese investors
Rainbow Medical, an Israeli-based seed investor and incubator specifically for medical device and medical technology startups, will open an office in Shanghai to help facilitate strategic deals as part of a $25 million cash infusion from investors in China.
February 19, 2015
Genmab and BioNovion expands their collaboration in the immune-oncology field
Genmab (Copenhagen, Denmark) and BioNovion (Oss, The Netherlands) have expanded their ongoing research agreement in immune-oncology to a full co-development and commercialisation deal in order to progress their bispecific antibody products that inhibit immune checkpoints.
Approval of drugs, vaccines, diagnostics and devices
February 13, 2015
US FDA approves Eisai new drug for thyroid cancer
US FDA approves Eisai’s kinase inhibitor Lenvima, or lenvatinib, to treat patients with progressive, differentiated thyroid cancer who are insufficiently responsive to radioactive iodine therapy.
U.S. orphan-drug approvals hit record high in 2014
An analysis of data from the FDA's Law Blog Orphan Drug Designations and Approvals database showed that the agency approved 49 orphan drugs last year, a 53% increase compared with 2013. The number of orphan-drug designations granted by the FDA's Office of Orphan Products Development also increased nearly 13% to 293, while the number of orphan-status requests reached 467, up almost 35% over 2013.
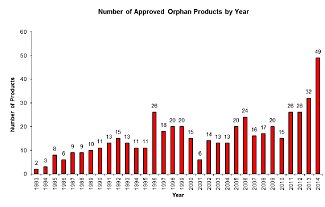
Orphan drug approvals climbed 53% to 49 in 2014–Courtesy FDA Law Blog
Nevertheless it is the very high price tag of orphan drugs that is going to pose hurdles for payers. A recent study indicated that orphan drugs could reach as much as 19% of new drug sales by 2020.
US FDA warns of device design amid UCLA superbug outbreak
FDA is warning doctors and hospitals that endoscope devices complex design may hinder proper cleaning contributing to the spread of the “drug resistant superbug” outbreak at the UCLA Health System in California. The agency's warning applies to duodenoscopes that are threaded through the mouth, throat, stomach or the top of the small intestine to drain fluids from blocked pancreatic or biliary ducts. Duodenoscopes' design makes the devices hard to sterilize, which could lead to broad transmission of infectious bacteria. The FDA warning comes a month after UCLA's Ronald Reagan Medical Center first reported deadly infections. Since then, seven patients were infected with a potentially deadly, drug-resistant strain of bacteria and Los Angeles public health authorities are tracking down at least 179 other patients who might have been exposed through contaminated scopes, according to the Washington Post article.
Drugs at clinical stage
February 19, 2015
US FDA grants priority reviews to Roche’s cobimetinib, a melanoma drug and for Pfizer Rapamune for rare lung disease
Roche gets a priority review for cobimetinib, a MEK inhibitor developed with Exelexis, in combination with Zelboraf (vemurafenib) for the treatment of patients with BRAF mutation-positive advanced melanoma.
The FDA grants Pfizer a Priority Review supplemental New Drug Application (sNDA) for the use of Rapamune (sirolimus) for the treatment of lymphangioleiomyomatosis (LAM), a rare, progressive often fatal lung disease in women that is characterized by the proliferation of disorderly smooth muscle growth (leiomyoma) throughout the lungs. The disease affects between 30,000 and 50,000 women worldwide. If Rapamune is cleared by the US FDA it will be the first approved treatment for the disease. Rapamune is an immunosuppressing drug already approved for organ rejection in kidney transplant patients.
The European Commission grants orphan status to aTyr Pharma’s muscular dystrophy drug
aTyr Pharma (San Diego, CA, USA) received orphan-drug status from the European Commission (EC) for Resolaris, a potential treatment for facioscapulohumeral muscular dystrophy (FSDH). Resolaris is an experimental first-in-class intravenous therapeutic protein developed by aTyr Pharma. FSDH is a rare and severe genetic myopathy with an immune component for which there is currently no approved treatment.
Miscellaneous
Concern about high prices of new cholesterol treatments
CVS Health is very concerned by the very high price of a coming class of cholesterol treatments, the PCSK9 drugs from Sanofi and Amgen, which are expecting FDA approval this summer. Both are biological drugs, with prices expected to reach $12,000 per year.
Former Shanghai official linked to GS bribery sentenced to 19 years in jail in China.
In December 2013, a few months after Chinese officials accused GSK of using travel agencies to bribe local doctors and healthcare professionals, Chinese police arrested Huang Fengping, former deputy director for the Shanghai Municipal Commission of Health and Family Planning. At the time, investigators found more than 400 envelopes stuffed with cash at his home, as well as gold bars and foreign currency in the boot of his car, the South China Morning Post reports. The Shanghai Number One Intermediate People's Court has jailed Fengping, for 19 years, according to a Xinhua report. The case is part of an ongoing country-wide anti-corruption initiative that has both involved foreign and domestic companies. In September 2014, Chinese authorities had already fined GSK $500 million, and former China GSK head Mark Reilly, reportedly received a three-year suspended sentence and deportation.
Author : Jean-Claude MULLER, Special Advisor, Innovation & International Relationship (I&IR)
Discover our services in Marketing & Business Development:
See All News
See other Biopharmaceutical News
see other Pharma & Biotech events in 2015
Last News
- US FDA Novel Drug Approval for 2025
- Update on Alzheimer’s disease treatments
- Le Populisme en France : Une note d’éclairage
Events
News archives
- January 2026
- December 2025
- November 2025
- September 2025
- July 2025
- June 2025
- May 2025
- April 2025
- February 2025
- January 2025
- December 2024
- November 2024
- October 2024
- September 2024
- August 2024
- July 2024
- June 2024
- March 2024
- February 2024
- January 2024
- November 2023
- September 2023
- July 2023
- April 2023
- March 2023
- January 2023
- December 2022
- November 2022
- October 2022
- August 2022
- June 2022
- May 2022
- April 2022
- March 2022
- January 2022
- December 2021
- November 2021
- October 2021
- September 2021
- August 2021
- July 2021
- June 2021
- May 2021
- April 2021
- March 2021
- February 2021
- January 2021
- December 2020
- November 2020
- October 2020
- September 2020
- July 2020
- June 2020
- May 2020
- April 2020
- March 2020
- February 2020
- January 2020
- December 2019
- November 2019
- October 2019
- September 2019
- June 2019
- May 2019
- April 2019
- March 2019
- February 2019
- January 2019
- December 2018
- October 2018
- June 2018
- May 2018
- March 2018
- February 2018
- January 2018
- December 2017
- November 2017
- October 2017
- September 2017
- August 2017
- July 2017
- June 2017
- May 2017
- April 2017
- March 2017
- February 2017
- January 2017
- December 2016
- November 2016
- September 2016
- July 2016
- June 2016
- May 2016
- April 2016
- March 2016
- February 2016
- January 2016
- December 2015
- November 2015
- October 2015
- September 2015
- August 2015
- July 2015
- June 2015
- May 2015
- April 2015
- March 2015
- February 2015
- January 2015
- December 2014
- November 2014
- October 2014
- September 2014
- June 2014
- May 2014
- April 2014
- March 2014
- January 2014
- November 2013
- September 2013
- July 2013
- May 2013
- April 2013
- March 2013
- January 2013
- December 2012
- November 2012
- October 2012
- March 2012





